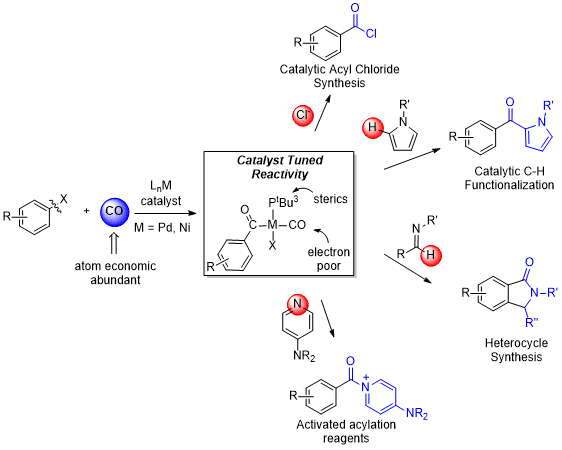
Transition metal complexes offer the ability to activate unreactive, readily available compounds towards chemical reactions. In this area, we are developing new classes of catalysts that can be used to transform carbon-halogen and carbon-hydrogen bonds into useful functionalized products. These hold the potential to streamline synthesis by obviating the functional group incorporation and manipulation steps typically needed to construct products.
For example, carbon monoxide is an attractive building block for organic synthesis as it both widely and inexpensively available and it is 100% atom economical. Late metal catalysts, in particular palladium, have been useful in this regard to prepare various carbonyl-containing derivatives. Our lab has focused on designing catalytic systems that use carbon monoxide to create highly reactive intermediates for their use in organic synthesis. This strategy allows access to products inaccessible by other carbonylative routes. This can include methods to incorporate aryl halides into heterocycles, and novel methods to directly functionalize C-H bonds in aromatic products. Moreover, the rational design of these catalysts also provides us with fundamental details on the mechanisms for these metal-catalyzed transformations allowing for the development of more active systems.